Human SAE1 Protein
SAE1, AOS1, SUA1, UBLE1A
- 100ug (NPP2478) Please inquiry
| Catalog Number | P13921-HNCB |
|---|---|
| Organism Species | Human |
| Host | Baculovirus-Insect Cells |
| Synonyms | SAE1, AOS1, SUA1, UBLE1A |
| Molecular Weight | The recombinant human CRIP2 consists of 348 amino acids and predicts a molecular mass of 38.6 KDa. It migrates as an approximately 39 KDa band in SDS-PAGE under reducing conditions. |
| predicted N | Gly |
| SDS-PAGE | 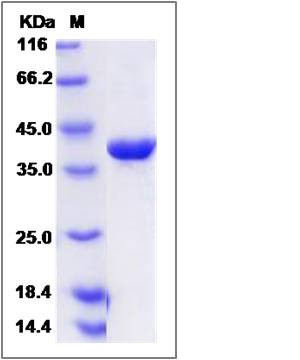 |
| Purity | > 95 % as determined by SDS-PAGE |
| Protein Construction | A DNA sequence encoding the human SAE1 (Q9UBE0) (Met1-Lys346) was expressed and purified. |
| Bio-activity | |
| Research Area | Immunology |Signal Transduction |ITIM/ITAM Immunoreceptors and Related Molecules |
| Formulation | Lyophilized from sterile 20mM Tris, 500mM Nacl, pH 7.4, 10% glycerol. 1. Normally 5 % - 8 % trehalose and mannitol are added as protectants before lyophilization. Specific concentrations are included in the hardcopy of COA. |
| Background | SAE1 belongs to the ubiquitin-activating E1 family. It is a heterodimer that acts as a E1 ligase for SUMO1, SUMO2, SUMO3, and probably SUMO4. It functions as a UBLI E1 ligase mediating the ATP-dependent activation of UBL1. SAE1 binds with UBLE1A and UBLE1B to form a heterodimer which can bind UBL1. SAE1 also regulates ATP-dependent activation of SUMO proteins and formation of a thioester with a conserved cysteine residue on SAE2. SAE1 and UBA2 form a heterodimer that functions as a SUMO-activating enzyme for the sumoylation of proteins. |
| Reference |
